program
GSRS23 web streaming link will be available on the GSRS23 event page of EFSA.
Please click the button below to join the meeting virtually.
All times are in Central European Summer Time (GMT+2)
All times are in Central European Summer Time (GMT+2)
Workshop I: “Workshop on Nanotechnology"
Tuesday, 26 September, 2023
THEME 1 | Qualification system supporting the use of NAMs in food
Chairs: Susanne Bremer-Hoffmann (EC JRC) & Anil Patri (US FDA)
14:00-14:05
Welcome and introduction to the event by the Chairs
Chairs
14:05-14:15
EMA Qualification of Novel Methodologies
14:15-14:25
US FDA qualification system for NAMs
14:25-14:35
EFSA NAMs4NANO Project
14:35-14:45
EFSA NAMs Case Study on Nanocellulose: the NANOCELLUP Project
14:45-15:35
Discussion
All
15:35-16:00
Coffee/Tea break
THEME 2 | Challenges and ways forward to investigate nanoplastics
Chair: Birgit Sokull-Kluettgen (EC JRC) & Anil Patri (US FDA)
16:00-16:05
Opening comments
Chairs
16:05-16:15
Presentation on the WHO report ‘Dietary and inhalation exposure to nano- and microplastic particles and potential implications for human health’
https://www.who.int/publications/i/item/9789240054608
16:25-16:35
Proxy measurands for nanoplastics – Should the development of analytical methods go in this direction?
16:35-16:45
Overview of ongoing activities involving micro/ nanoplastics from US NIST
16:45-17:25
Discussion
All
17:25-17:30
Concluding remarks
Chair
Poster session & Cocktail
Workshop II: Present and future of AI, open science and transparency in regulatory science
Tuesday, 26 September, 2023
SESSION 1 | New technologies and approaches in regulatory science
14:00-14:15
Welcome and introduction to the event by the chairs
Katya Tsaioun, EBTC (US), Federica Barrucci, EFSA, James Ramsay, EFSA
14:15-14:30
Big data/AI and their role in regulatory decision-making
14:30-14:45
Prerequisites for Data analytics and AI
14:45-15:00
The European ONTOX project: goals and first results
15:00-15:10
Q&A- identification/proposal of topics for group discussions
Chair
SESSION 2 | Breakout Group Discussions
15:10-15:50
Topic 1, 2, 3 group discussions (Heather chairs virtual)
Chairs: Katya Tsaioun EBTC, (US), Federica Barrucci, EFSA, James Ramsay, EFSA, Sebastian Hoffmann EBTC, Paul Whaley EBTC, Heather Schaefer, FDA (US)
15:50-16:05
Coffee break
SESSION 3 | Panel on open science, transparency and reproducibility needed in regulatory decisions
16:05-16:10
Reports back
Chairs (1 per group, 3 main points / important issues to work on)
16:10-16:40
Panel discussions
Thomas Margoni, KU Leuven (BE), Paul Whaley, Evidence-Based Toxicology (UK), Pinheiro Luis, EMA (NL), Pierre Eftekhari Inoviem (FR),
James Ramsay, EFSA
All times are in Central European Summer Time (GMT+2)
Day 1— Wednesday, 27 September, 2023
08:30-09:00
Registration
OPENING CEREMONY
09:00-09:30
Welcome and introduction to the event by the EFSA Chief Scientist
SESSION 1 | NAMs Global Landscape
Co-Chairs: Tucker Patterson, National Center for Toxicological Research (NTCR, FDA) (US) & Georges Kass (EFSA)
09:30-09:40
Opening Remarks
Co-Chairs
10:10-10:40
ToxAIcology—AI is the future of toxicology
Q&A
10:40-11:10
Coffee/Tea break
11:10-11:40
Bridging the divide between scientific development and regulatory application
Q&A
11:40-12:10
The Scientific Progress of Drug Regulation in China and its Regulatory Science Development
Q&A
12:10-12:40
NIHS 150th Anniversary and Regulatory Sciences
Q&A
12:40-14:00
Networking lunch
SESSION 2 | NAMs: Regulatory implementations
Co-Chairs: Suzanne Fitzpatrick, FDA (US) & Calvin Yeo, Singapore Food Agency (SFA) (SG)
14:00-14:10
Opening Remarks
Co-Chairs
14:10-14:30
Implementation of the 3RS at the EMA: past, present and future
Q&A
14:50-15:10
Activities of the Japanese Center for the Validation of Alternative Methods (JaCVAM), NIHS: Development of new toxicity tests using NAM and their use in regulations
Q&A
15:10-15:30
Regulatory landscape and critical Needs for NAMs in the Hazard assessment of industrial chemicals
Q&A
15:50-16:10
Coffee/Tea break
SESSION 3 | Regulatory Apps
Co-Chairs: Joanne Chan (Singapore Food Agency (SFA) (SG) and Catherine Carrillo Canadian Food Inspection Agency (CFIA) (CA)
16:10-16:20
Opening Remarks
Co-Chairs
16:20-16:40
Evaluation for developmental toxicity using human iPS cells
Q&A
16:40-17:00
Quality attributes and standards for mRNA-based and Lipid-based therapeutics
Q&A
17:00-17:20
How to handle human variability in risk assessment
Q&A
17:20-17:40
Case studies on refining risk assessment of food-related substances with NAMs
Q&A
17:40-18:00
FDALabel: A tool to facilitate regulatory application of drug labeling at FDA
Q&A
18:15-20:15
Poster session & Networking cocktail
All times are in Central European Summer Time (GMT+2)
Day 2—Thursday, 28 September, 2023
08:00-08:30
Registration
SESSION 4 | Emerging Technologies
Co-Chairs: Laila Mouawad Brazilian Health Regulatory Agency (Anvisa) (BR) & Yoko Hirabayashi National Institute of Health Sciences (NIHS) (JP)
08:30-08:40
Opening Remarks
Co-Chairs
09:20-09:40
Advancing Regulatory Science thru Innovation- Microphysiological Systems
Q&A
09:40-10:00
The challenge of Inter-Individual Variations for the standardisation of emerging technologies
Q&A
10:00-10:20
Safety assessment of cell-based therapeutic products derived from iPS cells
Q&A
10:20-10:40
Coffee/Tea break
SESSION 5| Artificial Intelligence /Machine Learning
Co-Chairs: Maurice Whelan, European Commission (EC, JRC) & Weida Tong, FDA (US)
10:40-10:50
Opening Remarks
Co-chairs
10:50-11:10
Newly developed artificial intelligence (AI) based bioimaging inspection system for lymph-node granuloma lesions in high-speed line slaughter plants
Q&A
11:10-11:30
Of Mice and Machines: Augmented Intelligence for Improving Chemical Safety Assessements
Q&A
11:50-12:10
AskYourDocs: harnessing the power of LLMs for a working cross-agency prototype on Information Retrieval from PDFs
Q&A
12:30-13:30
Networking lunch
SESSION 6 | Horizon scanning I
Co-Chair: Supriya Sharma Health Canada (CA) & Bo Li National Institutes for Food and Drug Control (NIFDC) (CN)
13:30-13:40
Opening Remarks
Co-Chairs
13:40-14:00
Structural analysis of therapeutic antibodies using CRYO EM
Q&A
14:00-14:20
An evidence pathway for trustworthy AI innovations: bridging the gap between developer and user communities
Q&A
14:40-15:00
Navigating Regulatory Genomics: Applications, Strengths, and Limitations in Monitoring Threats in the Food Supply Chain
Q&A
15:00-15:15
Short Coffee/Tea break
SESSION 7 | Horizon scanning II
Co-Chairs: Marta Hugas, former Chief Scientist, EFSA & Bill Slikker, former Director, NCTR, FDA (US)
15:15-15:25
Opening Remarks
Co-chairs
15:25-15:45
Innovation in Regulatory Science Awards at Burroughs Wellcome Fund —Preview of the Latest Technologies and Progress Towards Equitable Clinical Outcomes
Q&A
15:45-16:05
Toward social implementation and regulatory acceptance of MPS
Q&A
16:05-16:25
Development status and supervision of oragnoids and organ-on-a-chip in China
Q&A
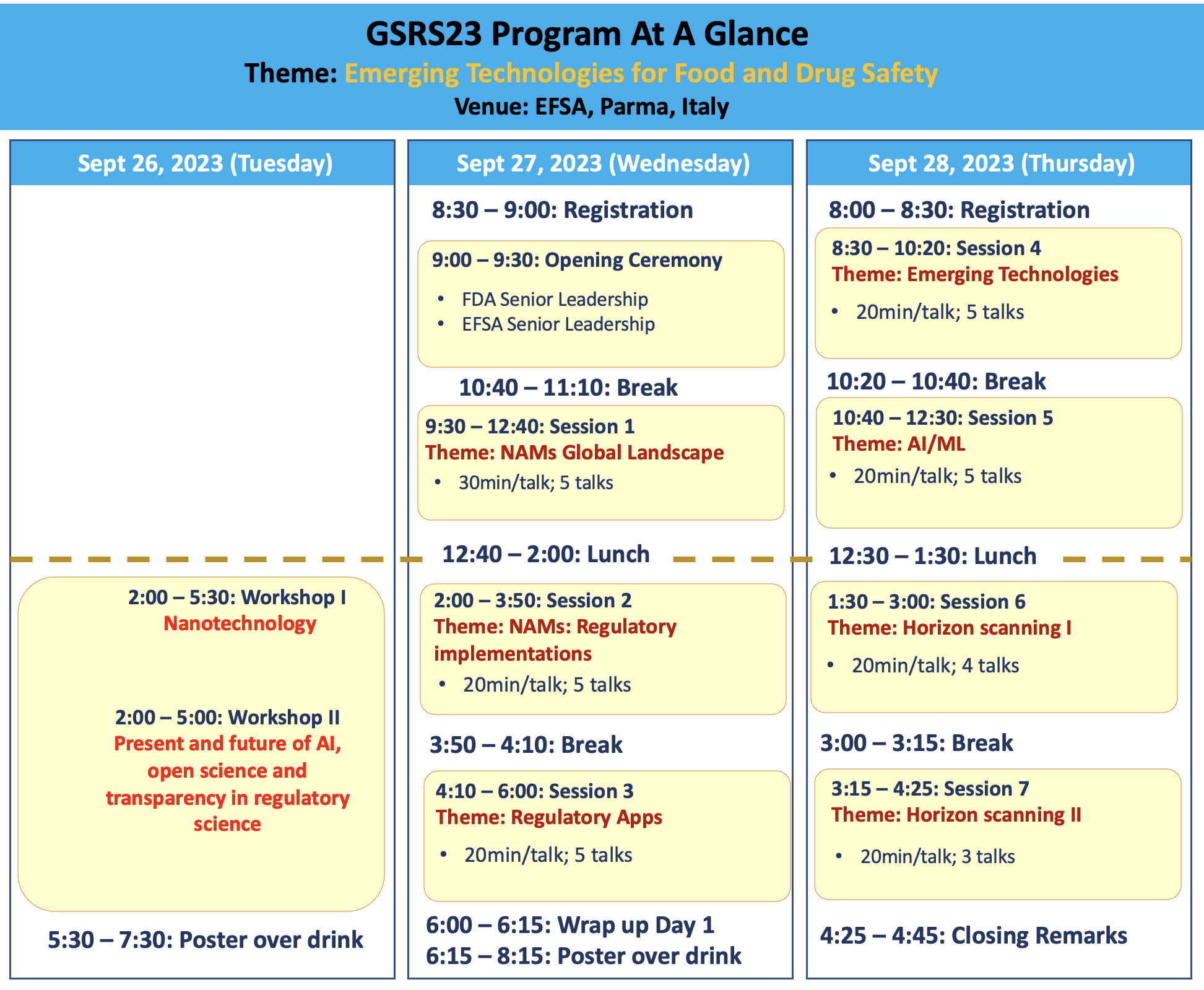
GSRS23 Program at Glance
APRIL Template
Support Center
Standard Support Enquiries
standard@april.com
Premium Support Enquiries
premium@april.com